 In order to decide, which type of flow is there, we should use the empirical equation called Reynold’s number. If the flow rate is higher and the diameter of the tube is large, then the flow becomes turbulent. Moreover, the diameter of the tube should be very small. We should keep in mind that, equation (4) applies to the liquids when the rate of flow is slow and steady. The measurement of viscosity of the liquid is based on Pioseulle’s equation (4) Measurement of Viscosity General Principle Remember that 1 pouse = 10 -1 kg m -1s -1. 
It means that a molecule of a liquid should gain sufficient energy to push aside the surrounding molecules before it can take part in the liquid flow.Ĭoefficients of Viscosity of Liquids at 20☌ in Poise and in SI Units (kg m -1 sec -1) It is the energy barrier that must be overcome before the flow can occur. The quantity ‘B’ is the activation of energy for- the viscous flow. The anomalous behavior of H 2O is seen from the graph because its hydrogen-bonded structure is broken with the rise in temperature. The following graphs show that many liquids show straight lines. From the slope of the straight-line ‘B’ can be calculated and from the intercept of the straight line factor, ‘A’ can be calculated. If we plot a graph between 1/T on the x-axis and log e 17 (in ŋ) on the y-axis, then a straight line is obtained. If we want to determine the constant ‘A’ and ‘B’ then we can convert it into the equation of a straight line by taking the natural log i.e. According to this equation, an increase of temperature decreases the viscosities of liquids. Mathematical Relationship between Viscosity and TemperatureĪrrhenius has given the following relationship between viscosity and temperature.Ī and B are the constants depending upon the nature of the liquid. It is estimated that for each one-degree rise in temperature, there is a 2% decrease in viscosity. In other words, a viscosity of a liquid falls with the increase in temperature. It means that a liquid starts flowing faster at high temperatures. The increase in temperature increases the kinetic energy of molecules of liquids. Try Also: What are Examples and Applications of Viscosity in Everyday Life? Effect of Temperature 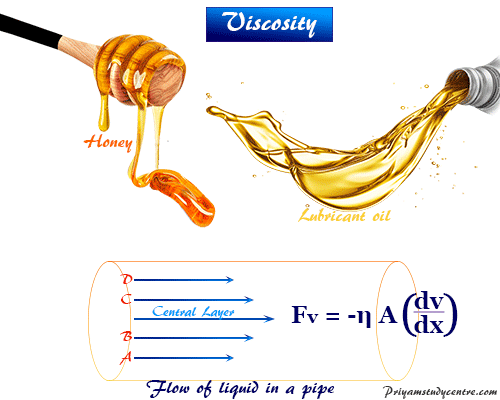
9 Examples of Viscosity in Everyday Life? The units of fluidity are reciprocal of viscosity i.e. “Fluidity is the reciprocal of viscosity.” “When a force of one dyne per square cm is maintained, between two layers which are 1cm apart and the difference of velocity between the two layers is 1 cm per second, then it is called poise”. The units of viscosity ”ŋ” can be derived from the equation (1) “It is the force per unit area, needed to maintain a unit difference of velocity between two parallel layers of the liquid, unit distance apart.” 
In the C.G.S system, the unit of “ŋ” is expressed as g cm -1 sec -1 and it is called Poise. It is also proportional to the area of the contact ‘A’ between the moving layers of the liquids. The force which is required to maintain the steady flow of liquid in direction of the force is directly proportional to the velocity gradient which is normal to the direction of flow. When the steady flow is reached then the velocity difference between any two layers becomes constant. When we move towards the center of the tube, then the velocities of the successive layers go on increasing. The layer adjacent to the walls of the tube is stationary. The liquid is considered to be arranged in a large number of parallel layers. In order to understand the viscosity, let us consider the flow of a liquid. It is the property that opposes the relative motion of adjacent layers. It means that different rates of flow of liquids depend upon a certain property and that is viscosity. Viscosity is a common observation that thick liquids like honey and mobile oil, flow slowly than many organic liquids like benzene and toluene, etc. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
